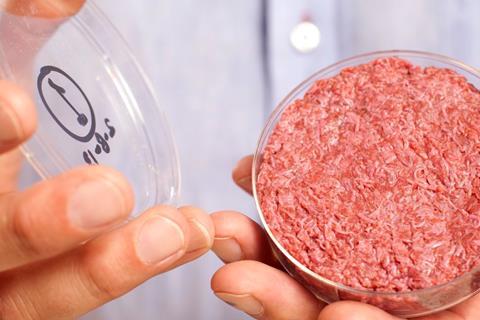
The government is to consider fast-tracking rules allowing food companies to sell products including lab-grown meats and insects, in a radical shake-up of food safety rules post-Brexit.
A new report by accountants Deloitte, commissioned by the FSA, found the move could help the UK meet its carbon reduction targets.
It comes amid fears that existing restrictions on so-called novel foods are holding back firms from investing in solutions to the environmental and hunger crisis.
An option flagged by the report suggests allowing alternative proteins to go on sale in the UK without the usual prolonged approvals process for novel foods, if they have been lawfully sold in other parts of the world.
Ministers included the potential to shake up rules on novel foods in a previous report called The Benefits of Brexit, published last year, which said the government wanted to take to take a “new approach” from the EU.
One of Deloitte’s most controversial suggestions is that the UK should adopt a system of “collaborative regulation” under which, instead of the existing approvals process which can take years, the FSA would authorise novel foods to be sold using the evidence base or decisions of food regulators in other countries.
It says this could also involve placing more responsibility on the food industry to “assure safety”.
The report says a single point of authorisation for novel foods is “not sustainable long-term” given the pace of innovation, and suggests a conditional authorisation and supervision model similar to that used in other sectors such as the pharmaceuticals industry.
However, it admitted the plans would require a “radical reimagining of the Novel Foods Regulatory Framework” and be based on greater consumer awareness of novel foods.
Supporters of such plans say Brexit could allow the UK to act to safeguard its food security by allowing products to come to market far quicker, and help to tackle future food supply chain crises caused by events such as the war in Ukraine.
They claim the red tape involved in gaining approval has acted as a barrier to investment.
The report suggests other potential solutions, including the FSA focusing more time on approvals for potential “high risk” foods and fast-tracking “lower risk” products.
The report comes with alternative foods gaining traction elsewhere in the world, especially in Asia and the US.
In November, Singapore became the first country to approve the sale of cultivated meat in the form of chicken nuggets and chicken breast, and in the same month the US Food & Drug Administration (FDA) gave approval for a lab-grown chicken product produced by Californian company Upside Foods.
“This report will be used as an input to support thinking within the FSA about how we design a future regulatory model, identifying the opportunities that make it easy for businesses to do the right thing and removing unnecessary barriers to innovation,” the FSA said in a statement yesterday.
It said it was committed to supporting innovation as well as safety.
Katrina Anderson, associate director at law firm Osborne Clarke LLP, said the report had huge implications for food companies in the UK.
“Regardless of which option is taken forward, the proposals amount to a radical shift in how novel foods would be approved which (if adopted) have the potential to allow companies to bring alternative proteins and other innovative foods to the UK market much more quickly.”



















No comments yet