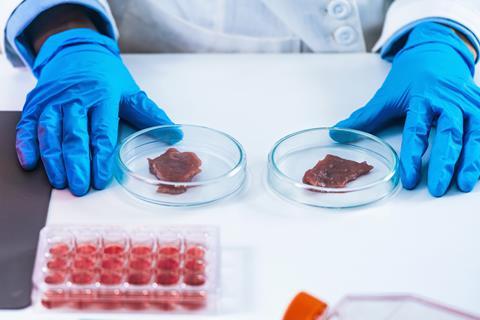
Plans for the UK to become the centre of a boom for lab-grown meat and other new novel foods are set to be dashed by the move to realign the UK with EU single market rules, The Grocer can reveal.
In October 2024, the Food Standards Agency predicted a rush of applications for cultivated cell-grown products after it revealed ministers had backed a fast-track system to prove they were safe.
The UK’s ability to set its own regulatory agenda for thousands of innovative new technologies previously bogged down in Brussels’ red tape was touted as one of the gains of the Brexit referendum result.
However, in its latest communication to manufacturers, it is understood the FSA has told companies they will have to return to the drawing board as a result of the under-negotiation Sanitary and Phytosanitary (SPS) deal with the EU.
There are fears the developments will see the so-called “sandbox” programme set up after a £1.6m grant from the Department for Science, Innovation & Technology become in the long run what one source described as a “huge white elephant”.
The FSA has billed the sandbox as a major economic, heath and environmental opportunity to develop foods that were beneficial for the environment that could eventually be tailored to improve the UK’s diet.
It said today that it would continue its work until being wound up in February next year.
Along with Food Standards Scotland, the FSA took responsibility for granting market authorisation for the sale of products such as food additives, food contact materials and food using new technology (such as lab-grown meat) after the UK left the EU.
However, the agency is now advising food companies that a return of the former EU regime is looming and warning that a “substantial” number of applications in the pipeline will not make it through before realignment.
The agency has written to around 600 market authorisation holders and businesses who have applications in its system, warning that dynamic alignment with EU law would mean that an EU market authorisation would in future be required to place a regulated food or feed product on the market. Any existing GB authorisations would therefore cease to apply.
“Given the proposed timescales for an agreement, a substantial number of applications in the GB Market Authorisation Service are unlikely to progress to the point of ministerial decision before the agreement is in place,” FSA market authorisation head of policy Rebecca Sudworth said.
“We have agreed with the board and with relevant ministers a set of prioritisation principles which we have applied to the current caseload, and we have communicated to applicants what this means for their applications.”
Lawyers told The Grocer the latest communications by the FSA made it clear that the prospect of the UK becoming the centre of a boom for new products such as lab-grown meat was effectively going to be killed off by the EU deal.
“An SPS agreement promises regulatory alignment with the EU which will enable food businesses to have consistent products on the market and labelling between GB and the EU,” said Katrina Anderson, a leading regulatory lawyer at Mills & Reeve.
“But it does also mean that all the post-Brexit efforts to streamline authorisations to bring innovative products to market potentially fall away.
“This would include products that might form part of the much-publicised cell-cultivated meat sandbox and precision fermentation and gene-edited foods, many of which have significant potential to make our diets more sustainable and provide better-tasting alternatives to meat and dairy.”
Anderson added: “There had been talk of exemptions for these products, but the latest from the government and FSA seems to suggest they are moving forward on the basis that there will not be exemptions.
“This is a blow to companies who have been developing innovative food products in the UK on the understanding that we would have a lighter touch route to market than the EU.”
The FSA denied that the investment in its sandbox and its wider programme on novel foods had been turned into a damp squib by the EU negotiations.
Deputy director for innovation Dr Thomas Vincent said: “Cell-cultivated products are a potential growth industry and these foods could have future benefits for food security and the environment.
“Our sandbox programme for cell-cultivated products remains on track and will run as planned until February 2027. The sandbox complements the market authorisation process by giving businesses a supported environment to develop their products, and it will continue to deliver that value throughout its duration.
‘The existing GB market authorisation process is still in place, and we are prioritising existing and new innovative food applications, including cell-cultivated products. We communicate regularly with applicants and any company with an application in the pipeline should remain in contact with us.”
He added: “The FSA will continue to provide the independent, evidence-based advice that businesses rely on, and we are committed to working closely with industry to support the safe introduction of these products to the market. We will keep businesses informed about the regulatory pathways as things progress.”



















No comments yet